

Kenya has a new contingency plan for tackling Rift Valley fever
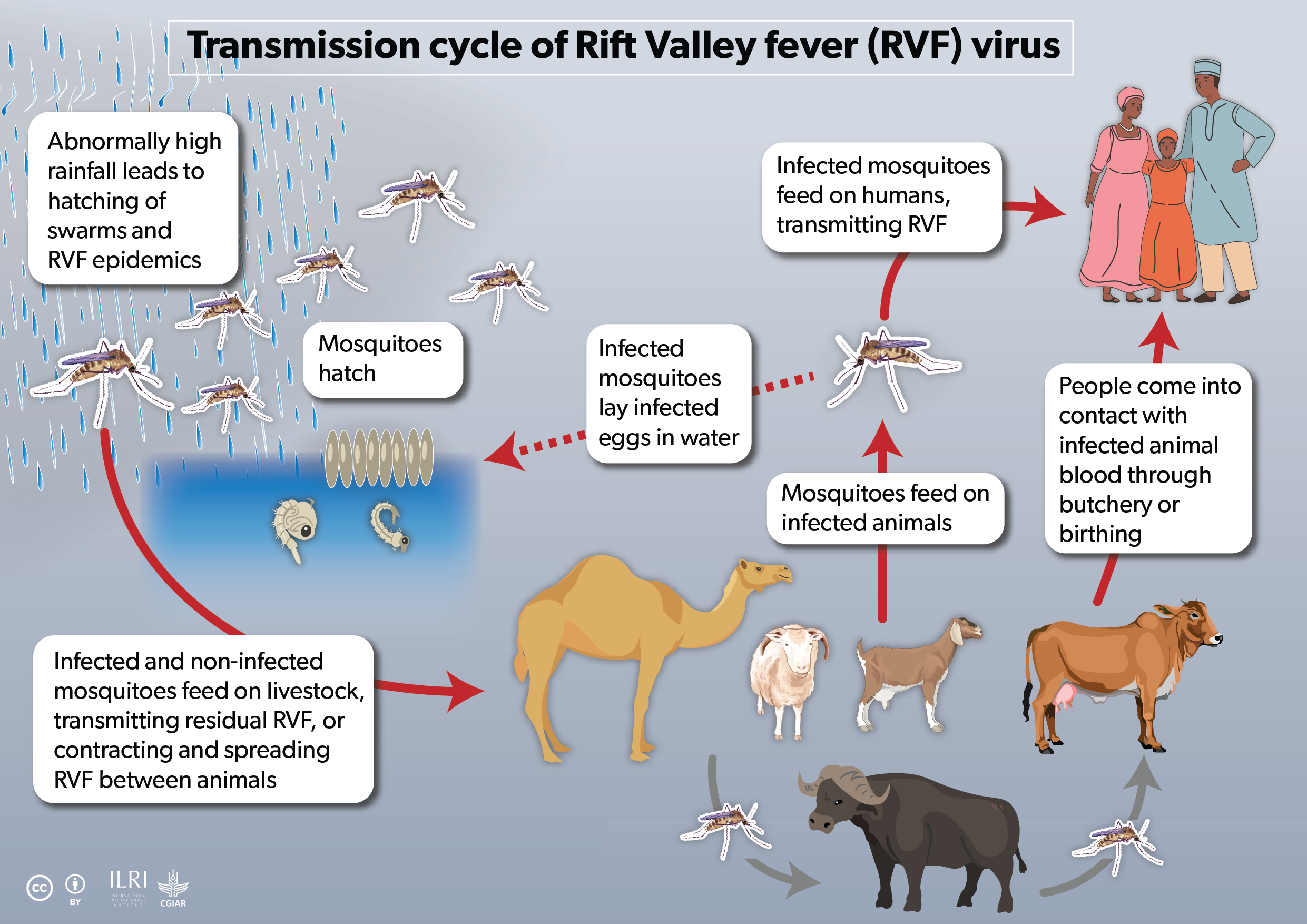
At the end of 2006, heavy rains battered East Africa, causing massive flooding. In the murky floodwaters, millions of mosquitos hatched—and hiding dormant in their eggs was the devastating Rift Valley fever Virus. As the clouds of insects fanned out to feed on warm-blooded animals, they spread the virus to sheep, cattle, goats, and camels.
The young were most affected. Lambs and kids died from internal bleeding. Cows and ewes spontaneously aborted their fetuses. And as pastoralists and veterinarians tried to help their animals, they caught the virus too. Across East Africa, 40,000 people are estimated to have gotten sick. In Kenya alone, 158 people died.
The economic effects were severe, too. In addition to the loss of livestock—a source of emergency cash, milk, food and livelihood for people in remote areas—the disease also shut down livestock trade to key markets in the Middle East, costing the country US$32 million in total. (Somalia fared even worse that year, with Rift Valley fever losses estimated at US$471 million dollars.)
“The disease causes havoc,” says Bernard Bett, a veterinarian and senior scientists at the International Livestock Research Institute (ILRI). And while subsequent epidemics in 2018 and 2024 weren’t as severe as the one in 2006, outbreaks are becoming increasingly frequent as climate change alters regional weather patterns.
In 2015, Kenya began work to update its Rift Valley fever contingency plan, aiming to improve the country’s response to the disease and minimise the impacts of future outbreaks. After a decade of research and fine-tuning by the Kenyan Government, ILRI, and other partners, in November 2025 the plan was finalised and implemented.
“This disease is one of the priority zoonotic diseases as a country that we deal with,” says Mathew Mutiiria - a medical epidemiologist from the Government of Kenya’s Zoonotic Disease Unit. “The contingency plan is a wake-up call for the government, and for our partners, to put our efforts together so as to adequately prepare for when the disease next appears.”
The first step, Mutiiria says, was work Bett’s team at ILRI did to pioneer a risk map for Kenya—identifying the specific counties that are at particularly high risk of Rift Valley fever outbreaks.
Most of these counties are in the country’s northeast, a generally arid area characterised by pastoral livestock systems, where herders move their animals seasonally from place to place. Counterintuitively, wetter areas are less affected by disease outbreaks, since livestock there live with mosquitos year-round, allowing them to build up immunity to the virus.
In the arid northeast, however, many animals only encounter mosquitos after unusually heavy rains, making them extremely vulnerable. Vaccination in these areas is also less effective, because the region’s severe droughts frequently wipe out entire herds, making it harder to sustain herd immunity. “If heavy rains and flooding come, you have a naive population—and explosions happen,” says Bett.
Knowing which counties are most at risk allows Kenya to focus its surveillance efforts, biosecurity equipment, training and preparation in a more cost-effective and targeted way, says Mutiiria—and can encourage at-risk counties to budget and plan for eventual outbreaks.
The contingency plan identifies several phases in disease control. Between epidemics, there’s a passive surveillance network in place. The government relies on tip-offs from vets, human health workers, and ‘community disease reporters’—village elders and other lay people tasked with keeping their eyes open for unusual events. “Because of the vastness of these areas and the few veterinarians, we really rely on the community,” says Khadija Chepkorir, a veterinarian from Kenya’s Zoonotic Disease Unit.
“They might report that there are a lot of lambs dying, so that becomes an early signal for us,” she says. So-called ‘abortion storms’ are another signal—“not just one female sheep aborting, but quite a number.”
It’s well-known that extreme climate events trigger Rift Valley fever outbreaks. Historically, epidemics have occurred after heavy rainfall in strong El Niño years, roughly once a decade. “El Niño can be predicted even six months down the line, and that was easy to know—if El Niño comes, you get Rift Valley fever,” says Bett. But climate change is messing with that pattern, and other environmental or livestock-management changes could also shift where and when outbreaks occur.
“Because of this climate variability, you may find an event occurring without prior alerts.”
However, if the climate and community clues do indicate an outbreak might be imminent, the government can implement active surveillance: sampling animals and mosquitos from at-risk areas for the presence of the virus. Counties can then use the advance notice to stock up on protective gear for vets and healthcare workers, and ramp up vaccination campaigns.
There is an effective lifetime animal vaccine for Rift Valley fever, which is made in Kenya at the country’s Veterinary Vaccine Production Institute. There are however several challenges in ensuring sufficient numbers of livestock receive it, Chepkorir says. Cost is a key factor—counties may not have the funds to buy and distribute enough doses through large and remote regions. (Because of gender dynamics, women-headed households are more likely to miss out.) Delivering Rift Valley fever vaccines alongside other animal vaccines such as those for peste des petits ruminants (PPR) or lumpy skin disease (LSD) could bring costs down, says Bett.
Unlike most Western countries, Kenya has not mandated livestock tagging and identification, he says. “It’s not part of the culture here yet.” That makes it impossible to identify which animals have already received a vaccine, causing inefficiencies.
Every year new animals are born, and must get their own vaccines to maintain herd immunity. And some farmers resist vaccinating their animals, fearing the jab will cause their animals to abort—a real but rare side effect which occurs much less frequently than abortions caused by the disease itself.
Following an outbreak, during the recovery phase, attention will shift to supporting farmers that have lost animals, quantifying the socioeconomic and health impacts of the disease—and assessing the nation’s performance against the contingency plan.
The fact that the document has been signed off and come into force sends a signal that the government takes Rift Valley fever very seriously, Mutiiria says. “But it's not enough for us to have the contingency plan in place. Now we have to put in the effort to ensure we actually do the activities that have been highlighted.”
That means running simulation exercises and trainings, carrying out surveillance, ramping up vaccination, and building capacity in regional laboratories that will test for the virus. “Above all, we need to have budgets set aside to mitigate the Rift Valley fever if it happens, especially in the areas where the risk map shows those high risks,” Mutiiria says.
Thanks to evidence-based research, collaboration and careful planning, when Rift Valley fever next strikes, Kenya will know what to do.

You may also like
Related Publications

Seasonal variation in mosquito abundance and environmental predictors in semi-pastoral southern Kenya: implications for endemic Rift Valley fever
- Gerken, Keli N.

Exploring gender and social vulnerabilities to Rift Valley fever: A socio-ecological One Health case study in Kenyan pastoralism
- Mutambo, Irene N.
Spatiotemporal patterns of Rift Valley fever virus in Africa: a retrospective genomic epidemiology and phylodynamic modelling study
- Juma, John

Hyperendemicity of Rift Valley fever in Southwestern Uganda associated with the rapidly evolving lineage C viruses
- Bakamutumaho, B.













